Singapore HSA Expands Chemiluminescence Reagent Regulation
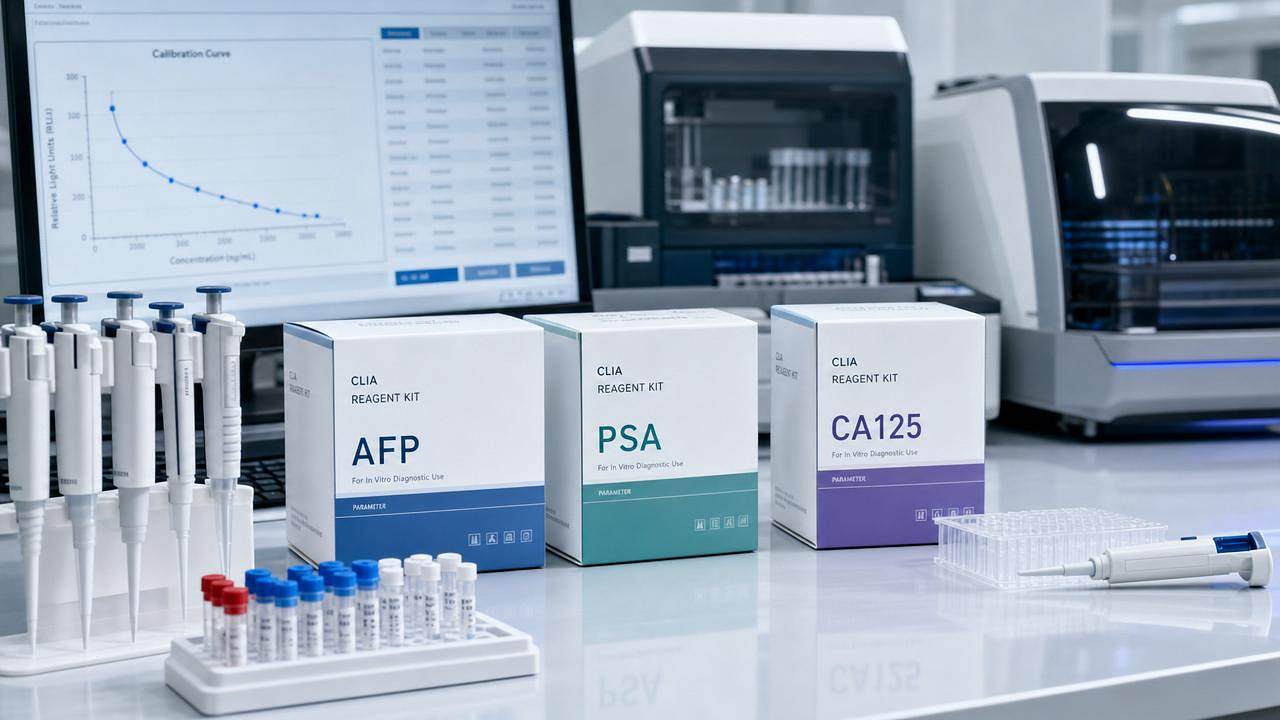
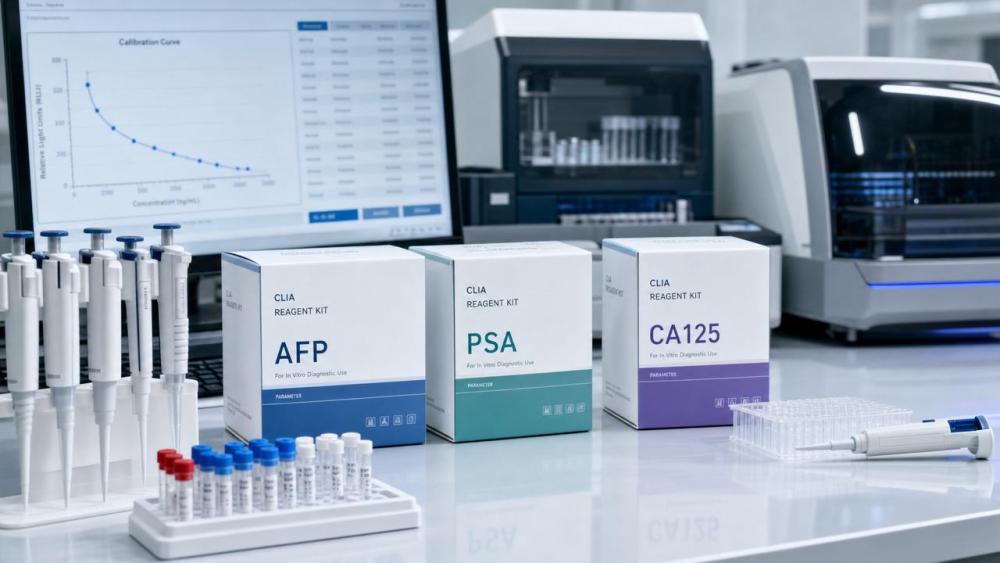
Singapore’s Health Sciences Authority (HSA) announced a significant regulatory update on 15 May 2026, expanding oversight of chemiluminescence-based immunoassay (CLIA) reagents—particularly those used for tumor marker detection. The change directly affects exporters, distributors, and service providers in the in vitro diagnostics (IVD) sector, especially those engaged in cross-border trade between China and Singapore. Its impact stems not from stricter registration requirements, but from newly mandated local clinical validation steps tied to Singaporean population baselines.
Event Overview
On 15 May 2026, the Singapore Health Sciences Authority (HSA) issued a notice extending its Chemical Luminescence-based Immunoassay Products (CEP) regulatory list to include 12 high-frequency tumor marker assays: AFP, CEA, CA125, PSA, CA19-9, CA15-3, NSE, SCC, CYFRA 21-1, β-hCG, Ferritin, and LDH. Effective 1 August 2026, all imported batches of these reagents must be accompanied by a ‘Local Population Baseline Comparison Report’—generated exclusively by HSA-designated laboratories in Singapore.
Industries Affected
Direct trading enterprises: Exporters and importers handling CLIA reagents for the listed tumor markers will face extended lead times (typically +4–6 weeks per batch) and added costs (~USD 1,800–2,500 per report). Since the report is batch-specific—not product-specific—repeat imports require repeat testing, undermining just-in-time supply models.
Raw material procurement enterprises: Firms sourcing critical components (e.g., monoclonal antibodies, magnetic beads, or chemiluminescent substrates) for tumor marker assays may experience upstream delays. Suppliers now need to align with downstream clients’ new documentation timelines; some are beginning to request pre-submission of lot-level stability and interference data to expedite local comparison workflows.
Manufacturing enterprises: Chinese IVD manufacturers producing CE-marked CLIA kits must now treat Singapore as a de facto clinical validation market—not merely a regulatory filing destination. While no new performance claims are required, the baseline report introduces an implicit requirement for analytical sensitivity/specificity alignment with local reference populations, prompting some firms to revisit assay calibration protocols ahead of shipment.
Supply chain service enterprises: Logistics and regulatory support providers offering ‘regulatory bridge’ services—including local representative appointments, customs clearance, and document verification—are seeing increased demand for end-to-end reporting coordination. Notably, only three Singapore-based labs are currently designated by HSA to issue the required reports, creating a capacity bottleneck and premium pricing for expedited slots.
Key Considerations and Recommended Actions
Verify designation status of intended testing labs
Confirm whether your chosen Singapore lab remains on HSA’s active list of designated facilities (updated quarterly); unlisted labs—even if ISO 15189-accredited—cannot issue valid reports.
Implement batch-level traceability and documentation planning
Integrate local comparison reporting into production scheduling: allocate ≥8 weeks between manufacturing completion and target Singapore arrival date, and retain full lot-level QC records (including calibrator/controls traceability) for potential audit review.
Evaluate feasibility of local sample pooling or collaborative studies
For firms exporting multiple related assays (e.g., full oncology panels), explore joint baseline studies across analytes—subject to HSA approval—to reduce per-assay reporting burden and cost.
Assess strategic value of establishing local technical representation
Companies without a Singapore entity may benefit from appointing a qualified local regulatory agent capable of liaising directly with HSA labs and managing report submission timelines—especially given limited lab capacity and strict report validity windows (6 months from issuance).
Editorial Perspective / Industry Observation
Observably, this policy shift reflects HSA’s broader pivot toward real-world clinical relevance over procedural compliance alone. Unlike EU IVDR’s focus on technical documentation depth, Singapore’s approach embeds local epidemiological context directly into market access—making it less about ‘what the test measures’ and more about ‘how it performs in our population’. Analysis shows this does not signal a move toward full local clinical trials, but rather a calibrated step toward evidence-based adoption thresholds. From an industry perspective, it is better understood not as a barrier, but as an early indicator of how ASEAN regulators may increasingly prioritize population-specific performance benchmarks—especially for high-impact diagnostic categories like oncology.
Conclusion
This regulatory expansion underscores a maturing regional approach to IVD oversight—one that balances global standards with localized clinical utility. For exporters, the immediate challenge lies in operational adaptation; for the broader IVD ecosystem, it signals a longer-term recalibration of what constitutes ‘market-ready’ in Southeast Asia. A rational interpretation is that such measures reward proactive engagement—not just regulatory literacy, but also clinical and logistical preparedness.
Source Attribution
Official notice: Singapore Health Sciences Authority (HSA), ‘Notice on Expansion of CEP Regulatory Scope’, Ref. No. HSA/IVD/NOTICE/2026/007, published 15 May 2026. Available at: https://www.hsa.gov.sg/ivd-notices.
Note: HSA has indicated that the list of designated laboratories and acceptable report templates will be updated on a rolling basis; stakeholders are advised to monitor HSA’s IVD portal for revisions through Q2 2026.
Recommended News
Weekly Insights
Stay ahead with our curated technology reports delivered every Monday.