Healthcare intelligence is reshaping smarter care decisions
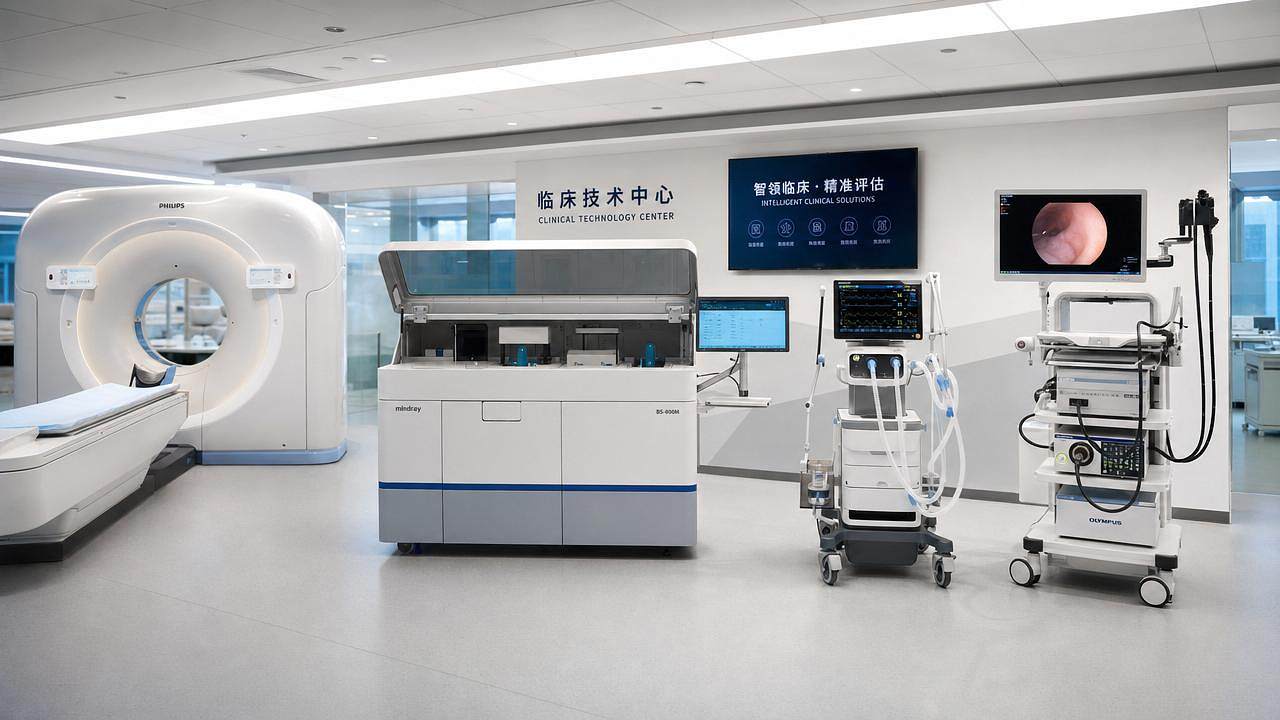
Healthcare intelligence is reshaping smarter care decisions by turning complex imaging, IVD, life support, surgical, and endoscopic data into actionable clinical and investment insight. For technical evaluators, AMDS connects performance, compliance, and precision medicine requirements, helping identify technologies that deliver measurable diagnostic value, safer workflows, and stronger long-term ROI in a rapidly evolving global MedTech landscape.
For buyers and technical review teams, the challenge is no longer access to information. It is filtering signal from noise across 5 critical equipment domains, 3 decision layers, and multiple regulatory pathways. A scanner may offer excellent image sharpness, but if reconstruction speed, service uptime, or CE MDR documentation are weak, the investment case quickly erodes.
This is where healthcare intelligence becomes operational rather than conceptual. It helps evaluators compare clinical fit, workflow consequences, data quality, interoperability, and cost exposure before procurement moves into tender, pilot, or deployment. In high-stakes MedTech environments, good intelligence shortens evaluation cycles, reduces technical blind spots, and supports safer long-term adoption.
Why healthcare intelligence matters across modern clinical equipment categories
AMDS focuses on 5 pillars that define the upper limit of modern hospital capability: medical imaging, IVD, life support, core operating room systems, and medical endoscopy. Each pillar generates different forms of evidence, from image resolution and assay sensitivity to ventilation reliability and optical stability. Healthcare intelligence turns those disconnected data points into comparable decision criteria.
For technical evaluators, the real question is not whether a device is advanced. It is whether the device performs consistently under clinical workload, integrates into existing infrastructure within 2 to 12 weeks, and supports compliance expectations in target markets such as FDA-regulated or CE MDR-governed regions.
Five equipment domains require five different evaluation logics
A CT system may be judged by detector architecture, dose management, reconstruction latency, and throughput per hour. An IVD analyzer is more often assessed on test menu breadth, sample-to-result time, reagent stability, contamination control, and LIS connectivity. A ventilator or ECMO platform requires a stronger emphasis on fail-safe logic, alarm hierarchy, uptime resilience, and maintenance intervals.
Similarly, operating room infrastructure is evaluated through ergonomics, positioning range, lighting consistency, and cleaning compatibility. Endoscope systems demand close scrutiny of 4K or 3D visualization quality, anti-fog behavior, tip maneuverability, sterilization workflow, and image persistence under fluid-heavy procedures lasting 60 to 180 minutes.
Typical evidence types reviewed during technical assessment
- Performance data: resolution, sensitivity, response time, uptime, image or assay repeatability
- Workflow data: setup time, case turnover time, user interface complexity, training requirements
- Compliance data: technical files, risk controls, audit readiness, labeling, post-market expectations
- Financial data: service burden, consumable dependence, spare parts cycle, reimbursement alignment
The table below shows how healthcare intelligence supports technical comparison across the main clinical equipment categories AMDS tracks. It highlights the operational metrics that most often influence shortlist decisions.
The key takeaway is that healthcare intelligence improves procurement only when it is category-specific. A uniform scorecard rarely works across all 5 domains. AMDS addresses this by linking engineering details, compliance readiness, and commercial practicality into a single evaluation narrative that technical teams can defend internally.
From raw device data to decision-grade intelligence
In most hospitals and MedTech organizations, technical review suffers from fragmented evidence. Clinical teams want outcome relevance. Engineering teams want parameter validation. Procurement wants lifecycle visibility. Finance wants ROI under reimbursement pressure, especially where DRG-based payment models shape capital spending over 3 to 7 years.
Healthcare intelligence bridges these requirements by stitching technical, regulatory, and economic inputs. Instead of reviewing image quality separately from market access or serviceability, evaluators can assess the full operational picture: how fast a system installs, how often it needs intervention, what training depth is required, and where utilization thresholds begin to justify the investment.
How technical evaluators should assess smarter care technologies
A strong evaluation framework usually includes 4 layers: clinical value, technical performance, compliance readiness, and economic sustainability. Healthcare intelligence strengthens all 4 by turning isolated product claims into structured comparisons. This is especially important when multiple vendors appear similar on brochures but diverge significantly in real-world deployment conditions.
AMDS supports this process by examining equipment from the inside out. Image reconstruction algorithms, biomarker detection pathways, anti-fog optical design, and ICU reliability logic must all be understood at the mechanism level before they can be judged at the procurement level.
Step-by-step framework for technical evaluation
- Define use case boundaries: screening, acute care, minimally invasive surgery, or high-volume routine diagnostics.
- Set 6 to 10 measurable criteria such as turnaround time, image consistency, sensitivity threshold, uptime target, or training time.
- Check regulatory and documentation readiness for target markets within the next 12 to 24 months.
- Model service, consumables, and utilization economics across expected case volume.
- Run pilot validation with cross-functional stakeholders before final sourcing.
What each step should answer
Step 1 clarifies whether the product solves a narrow or broad problem. Step 2 protects evaluators from vague claims. Step 3 prevents hidden access delays. Step 4 reveals lifecycle burden. Step 5 tests whether the theoretical value survives in day-to-day clinical operations. In practice, missing any one of these 5 steps can delay implementation by 4 to 16 weeks.
The next table provides a practical procurement-oriented checklist. It can be adapted for imaging systems, IVD analyzers, life support devices, or endoscopic platforms where smarter care decisions depend on more than one technical dimension.
Used correctly, this checklist prevents one of the most common procurement errors: selecting on visible specification strength while ignoring invisible execution risk. Healthcare intelligence is valuable because it makes those hidden variables visible before purchasing commitments harden.
Common mistakes in MedTech evaluation
Technical evaluators often face pressure to accelerate decisions. Under time constraints, three mistakes appear repeatedly. First, teams over-prioritize peak performance instead of stable performance across 8-hour or 24-hour care cycles. Second, they underestimate service architecture, including spare parts access and remote troubleshooting readiness. Third, they separate compliance review from technical review, which can create painful surprises later.
- Do not score innovation without scoring maintainability.
- Do not assume software maturity from interface polish alone.
- Do not evaluate high-end imaging ROI without utilization assumptions.
- Do not approve ICU-critical equipment without failure-mode review.
Why compliance intelligence changes the buying outcome
For exported medical equipment, compliance is not a final checkpoint. It is a design and commercialization constraint from day 1. CE MDR and FDA expectations can influence labeling structure, evidence quality, software documentation depth, and post-market surveillance planning. A technically promising device may still be commercially delayed if these elements are weak.
This is one of AMDS’s strongest value areas. By combining compliance analysis, engineering interpretation, and health economics review, the platform gives technical evaluators a more complete basis for decisions. That matters whether the goal is hospital adoption, international market entry, or internal product positioning against established competitors.
Applying healthcare intelligence to ROI, implementation, and long-term value
Smarter care decisions are not limited to diagnosis quality. They also involve throughput, staffing efficiency, risk reduction, and budget resilience over time. In many settings, a device that costs more upfront may create stronger value if it reduces repeat scans, lowers manual intervention, shortens turnaround, or supports more cases per shift.
Healthcare intelligence helps evaluators quantify that tradeoff. Instead of asking whether a premium system is expensive, the better question is whether its total operating profile supports measurable gains over 36 to 60 months. This is especially relevant for high-end imaging, molecular diagnostics, and critical care equipment where downtime or weak performance carries disproportionate clinical cost.
Three ROI lenses that matter in clinical MedTech
The first lens is utilization. Can the system handle enough cases per day to justify its footprint and service contract? The second is diagnostic or procedural value. Does it improve confidence, reduce repeats, or support earlier intervention? The third is operational resilience. Does it sustain performance under volume stress, staffing variation, and maintenance constraints?
Under DRG-style payment pressure, hospitals increasingly assess capital equipment through these 3 lenses rather than through acquisition price alone. A 10% improvement in throughput or a reduction in avoidable rework may matter more than a lower initial quote if the equipment serves a high-volume service line.
Implementation planning for technical teams
Once a technology is selected, implementation discipline becomes decisive. In typical deployments, technical teams should plan around 4 phases: pre-install assessment, infrastructure preparation, acceptance validation, and post-go-live optimization. Depending on device complexity, these phases may span 2 weeks for a modest analyzer upgrade or 8 to 16 weeks for advanced imaging or integrated OR systems.
Each phase should include clear ownership. Biomedical engineering may own site readiness. Clinical users may define acceptance scenarios. IT may govern network and system integration. Vendor teams may handle installation, calibration, and core training. Healthcare intelligence supports this phase by identifying likely bottlenecks before the implementation clock starts.
Post-deployment indicators worth tracking
- Utilization rate in the first 30, 90, and 180 days
- Repeat exam or repeat test frequency
- Downtime hours per month and average response time
- Training completion rate across relevant user groups
- Consumable variance against forecast
These indicators make healthcare intelligence continuous rather than one-time. They also create a feedback loop for future procurement decisions, helping technical evaluators refine specifications, supplier expectations, and deployment standards across the organization.
Where AMDS creates practical value for evaluators
AMDS is positioned as a strategic intelligence center for clinical MedTech, especially where advanced engineering intersects with strict compliance and precision medicine demands. Its value lies in connecting details that are often evaluated separately: AI-assisted image reconstruction, molecular detection logic, critical-care reliability, endoscopic optical performance, and reimbursement-driven investment logic.
For technical assessment teams, that means better visibility into what truly drives clinical safety, frontier efficacy, and commercial readiness. Rather than relying on isolated vendor narratives, evaluators can use healthcare intelligence to build a defensible comparison framework, reduce procurement risk, and align technology choice with both operational need and strategic growth.
In a market where advanced devices are becoming more digital, more connected, and more specialized, healthcare intelligence is no longer optional. It is the discipline that helps technical evaluators translate complexity into confident action across imaging, IVD, life support, operating room systems, and minimally invasive surgery platforms.
AMDS supports that discipline by combining engineering insight, compliance interpretation, and health economics logic into one decision-ready perspective. If your team is comparing new clinical technologies, preparing for market access, or validating long-term ROI, now is the time to get a more precise evaluation framework in place. Contact AMDS to explore tailored intelligence support, request a customized assessment approach, or learn more about solution pathways for smarter care decisions.
Recommended News
Related News
- 00
0000-00
Digital healthcare solutions can fail without this plan - 00
0000-00
How to compare advanced diagnostic systems before buying - 00
0000-00
Healthcare intelligence is reshaping smarter care decisions - 00
0000-00
How outdated hospital technology infrastructure slows care - 00
0000-00
What is driving medical equipment innovation in 2026?
Weekly Insights
Stay ahead with our curated technology reports delivered every Monday.