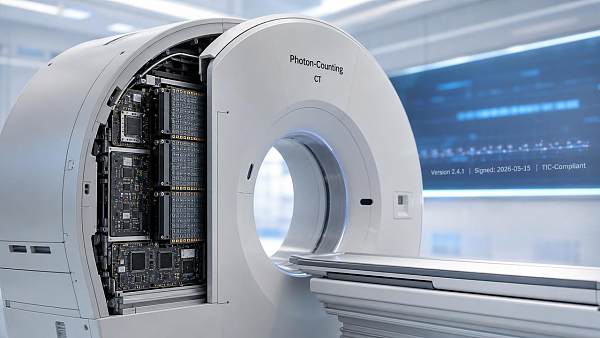
EU Launches Photon-Counting CT Traceability Pilot
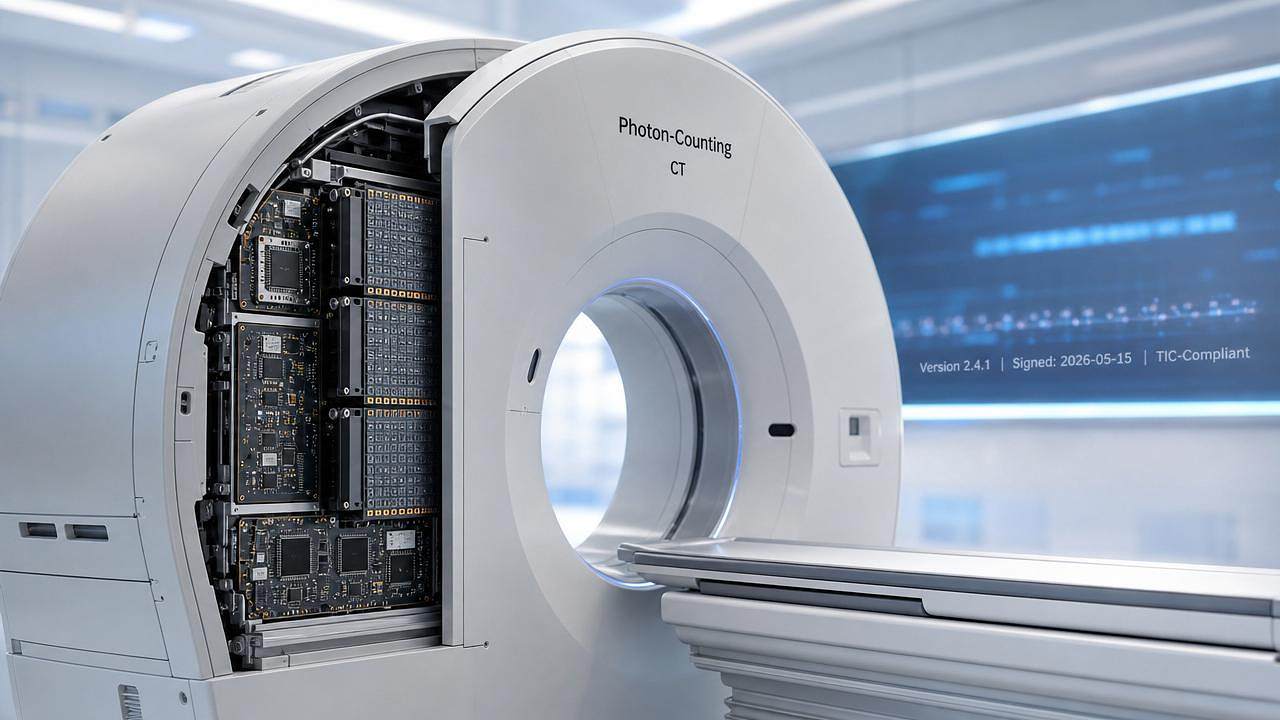
The European Commission, in collaboration with TÜV Rheinland, launched the Traceable Imaging Chain (TIC) pilot for photon-counting CT systems on May 15, 2026 — a regulatory initiative requiring verifiable digital signatures and version traceability across 17 critical nodes in the clinical imaging chain. This development is especially relevant for medical imaging equipment manufacturers, AI algorithm developers, regulatory affairs teams, and global procurement stakeholders in diagnostic radiology.
Event Overview
On May 15, 2026, the European Commission and TÜV Rheinland jointly initiated the Traceable Imaging Chain (TIC) pilot project for photon-counting CT systems. The program mandates that device manufacturers provide auditable, digitally signed version records for 17 defined imaging chain components — including detectors, ASIC chips, reconstruction algorithms, and calibration protocols. Three Chinese suppliers — United Imaging Healthcare, Neusoft Medical, and Shanghai United Imaging Intelligent Healthcare — have been included in the first mutual recognition directory.
Industries Affected by Segment
Medical Imaging Equipment Manufacturers
Manufacturers supplying photon-counting CT systems to the EU market will face new technical documentation and version-control obligations. Impact manifests in increased pre-market validation effort, mandatory integration of cryptographic signing into firmware/software release pipelines, and potential redesign of QA workflows to support node-level auditability.
AI Algorithm Developers & Software Vendors
Reconstruction algorithms are explicitly listed as one of the 17 traceable nodes. Developers must now maintain immutable version logs, provenance metadata, and signature-capable deployment mechanisms — not just for model weights, but for full inference pipelines (including preprocessing, normalization, and post-processing steps).
Regulatory Affairs & Compliance Service Providers
Firms supporting CE marking or EU MDR submissions will need to adapt technical file templates to include TIC-specific evidence: digital signature schemes, timestamped version archives, and cross-node consistency verification reports. This adds a new layer beyond current Annex II requirements.
Global Procurement & Health Technology Assessment Bodies
Hospitals and national procurement agencies evaluating photon-counting CT systems may begin incorporating TIC compliance as a formal criterion in tender specifications. Long-term trust assessments — particularly around algorithmic stability and field-upgrade accountability — are now formally linked to traceability infrastructure.
What Relevant Enterprises or Practitioners Should Focus On
Monitor official guidance from the European Commission and TÜV Rheinland
The TIC pilot is currently a voluntary phase; however, analysis shows that its design principles — especially algorithm versioning and cryptographic signing — are likely to inform future revisions of the EU MDR Annex I general safety and performance requirements.
Identify and map internal imaging chain nodes against the published list of 17
Current more actionable than broad strategy is to conduct an internal inventory: which hardware modules, firmware versions, algorithm binaries, and calibration SOPs fall within the defined scope — and whether existing build, release, and archiving systems support cryptographic signing and long-term verifiability.
Distinguish between policy signal and operational mandate
Observably, the pilot does not yet constitute a legal requirement for CE marking. However, inclusion in the mutual recognition directory signals early alignment with emerging expectations — making participation a de facto benchmark for commercial credibility among EU health technology buyers.
Prepare documentation and communication protocols for upstream/downstream traceability
Suppliers of detectors or ASICs should anticipate requests for signed version attestations from system integrators. Similarly, hospitals may request signed lineage reports for installed systems — meaning supply chain coordination and API-ready audit logs are becoming operationally relevant now.
Editorial Perspective / Industry Observation
This initiative is better understood as a regulatory signal than an immediate compliance threshold. Analysis shows it reflects a broader shift: the EU is treating algorithmic behavior not as a black-box output, but as a chain of accountable engineering decisions — each subject to version control, verification, and lifecycle governance. It signals growing emphasis on ‘algorithmic stewardship’ as a core dimension of clinical device trustworthiness. While full standardization remains years away, early adopters gain visibility into evolving expectations — particularly around transparency in AI-augmented reconstruction.
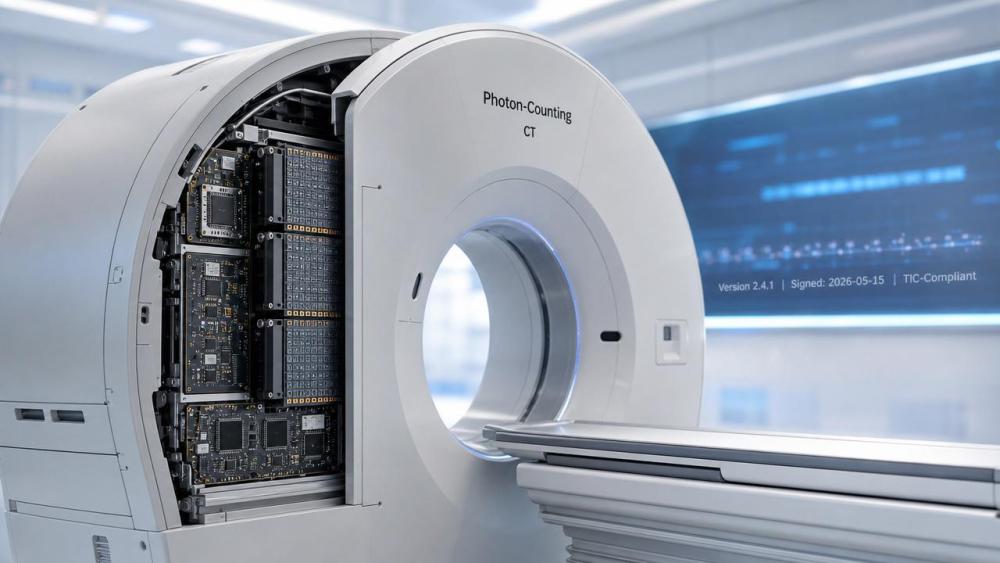
Conclusion: The TIC pilot marks a procedural inflection point — not a sudden regulatory barrier, but a calibrated step toward embedding verifiability into high-end medical imaging infrastructure. For industry actors, it is more productively viewed as a forward-looking framework for quality assurance than a near-term certification hurdle. Current interpretation should emphasize preparedness over panic, documentation over disruption, and interoperable traceability over proprietary silos.
Source: European Commission press release (May 15, 2026); TÜV Rheinland TIC pilot documentation (publicly announced May 2026).
Note: The scope of future regulatory adoption — including potential extension to other modalities or mandatory status — remains under observation and has not been confirmed.
Recommended News
Related News
- 00
0000-00
EU Launches Photon-Counting CT Traceability Pilot - 00
0000-00
TÜV Rheinland Launches First Photon-Counting CT Imaging Chain Certification - 00
0000-00
TÜV Rheinland Launches First PC-CT Imaging Chain Certification - 00
0000-00
Medical Imaging Diagnostics Choice Gets Harder as CT Advances - 00
0000-00
Is Photon-Counting CT Worth the Upgrade for Imaging ROI?
Weekly Insights
Stay ahead with our curated technology reports delivered every Monday.